Difference between revisions of "Main Page"
(→Workshops) |
(→Announcements) |
||
| Line 4: | Line 4: | ||
== Announcements == | == Announcements == | ||
| + | |||
| + | ===Molecular Dynamics=== | ||
| + | ==== SARS-CoV-2 D614 Spike Cleavage Site Interaction with Furin ==== | ||
| + | <html> | ||
| + | <div style="text-align:center;"> | ||
| + | <video width="700" height="500" controls autoplay loop> | ||
| + | <source src="https://crtc.cs.odu.edu/assets/videos/S5-D614-w-furin.mp4" type="video/mp4"> | ||
| + | Your browser does not support the video tag. | ||
| + | </video> | ||
| + | </div> | ||
| + | </html> | ||
| + | |||
| + | ==== SARS-CoV-2 G614 Spike Cleavage Site Interaction with Furin ==== | ||
| + | <html> | ||
| + | <div style="text-align:center;"> | ||
| + | <video width="700" height="500" controls autoplay loop> | ||
| + | <source src="https://crtc.cs.odu.edu/assets/videos/S6-G614-w-furin.mp4" type="video/mp4"> | ||
| + | Your browser does not support the video tag. | ||
| + | </video> | ||
| + | </div> | ||
| + | </html> | ||
| + | |||
===Molecular Geometric Feature Analysis=== | ===Molecular Geometric Feature Analysis=== | ||
Revision as of 21:36, 5 November 2020
Contents
Announcements
Molecular Dynamics
SARS-CoV-2 D614 Spike Cleavage Site Interaction with Furin
SARS-CoV-2 G614 Spike Cleavage Site Interaction with Furin
Molecular Geometric Feature Analysis
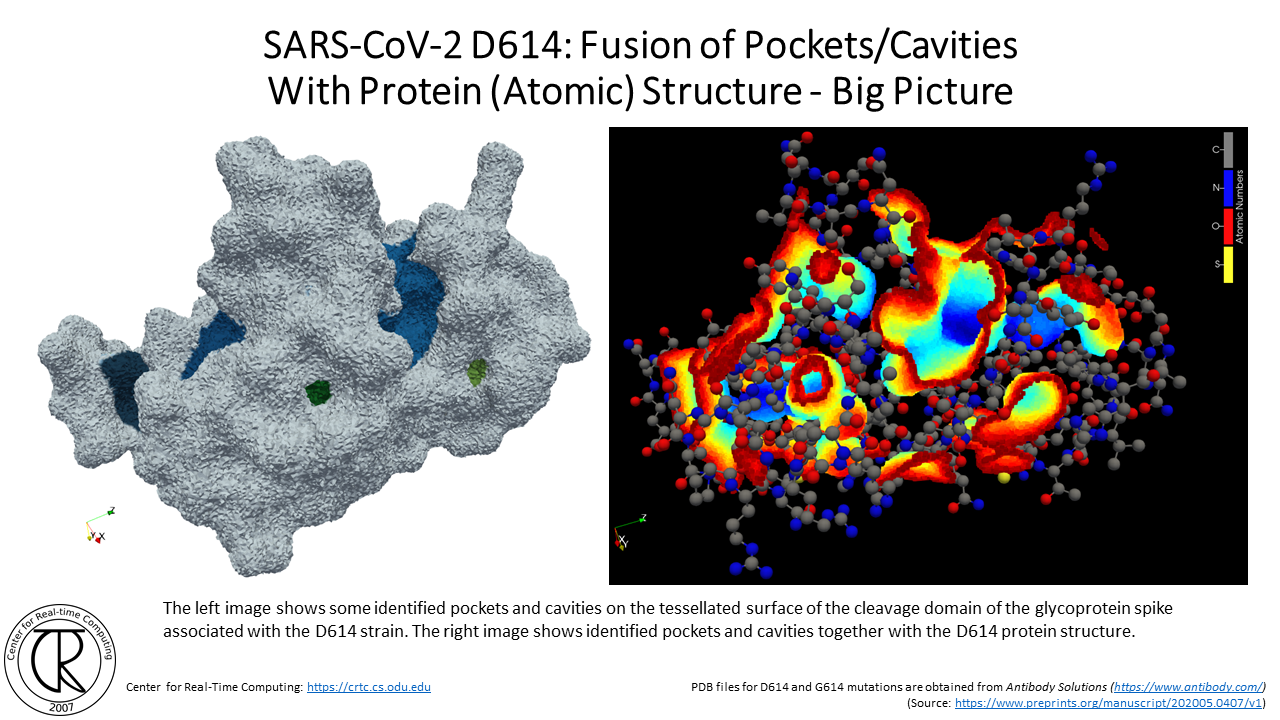
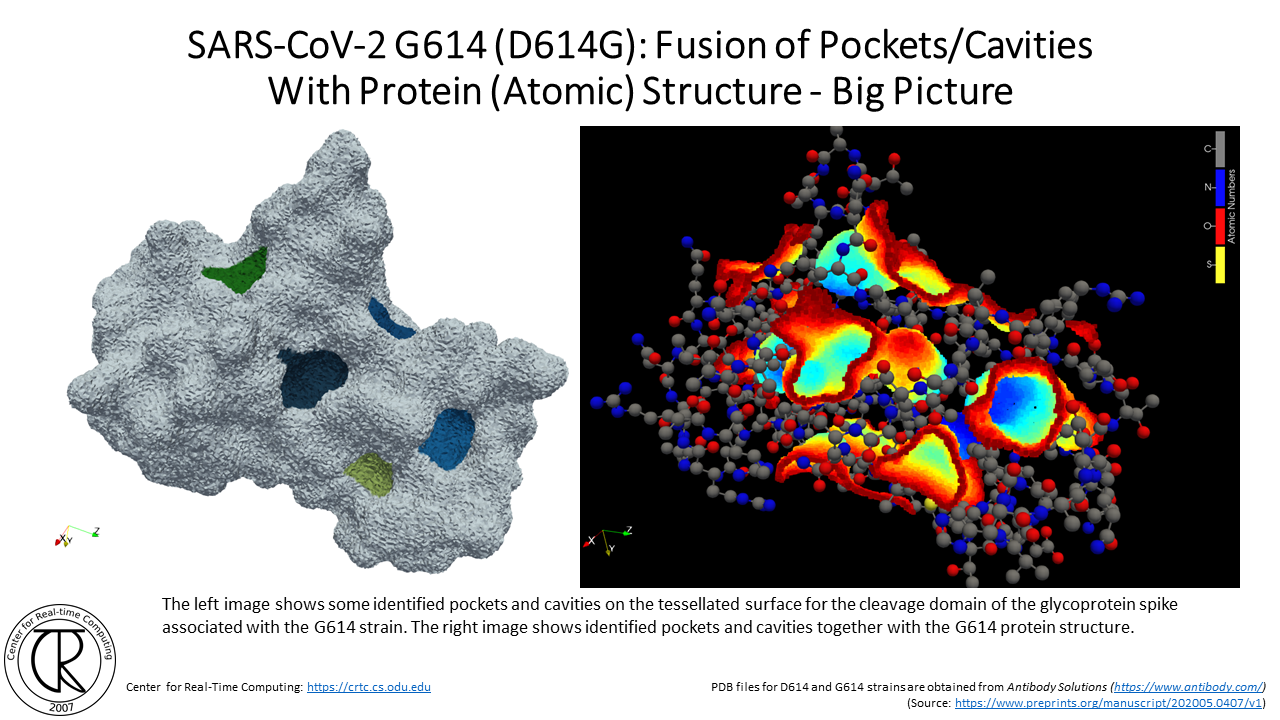
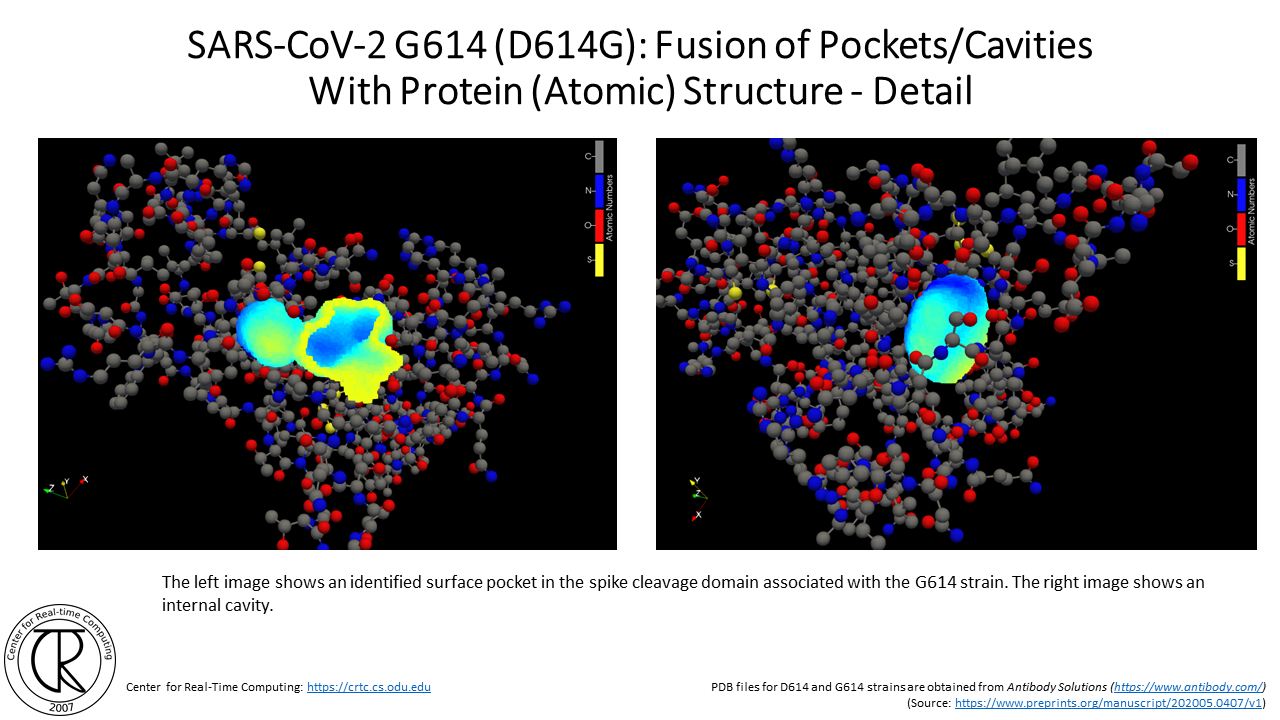
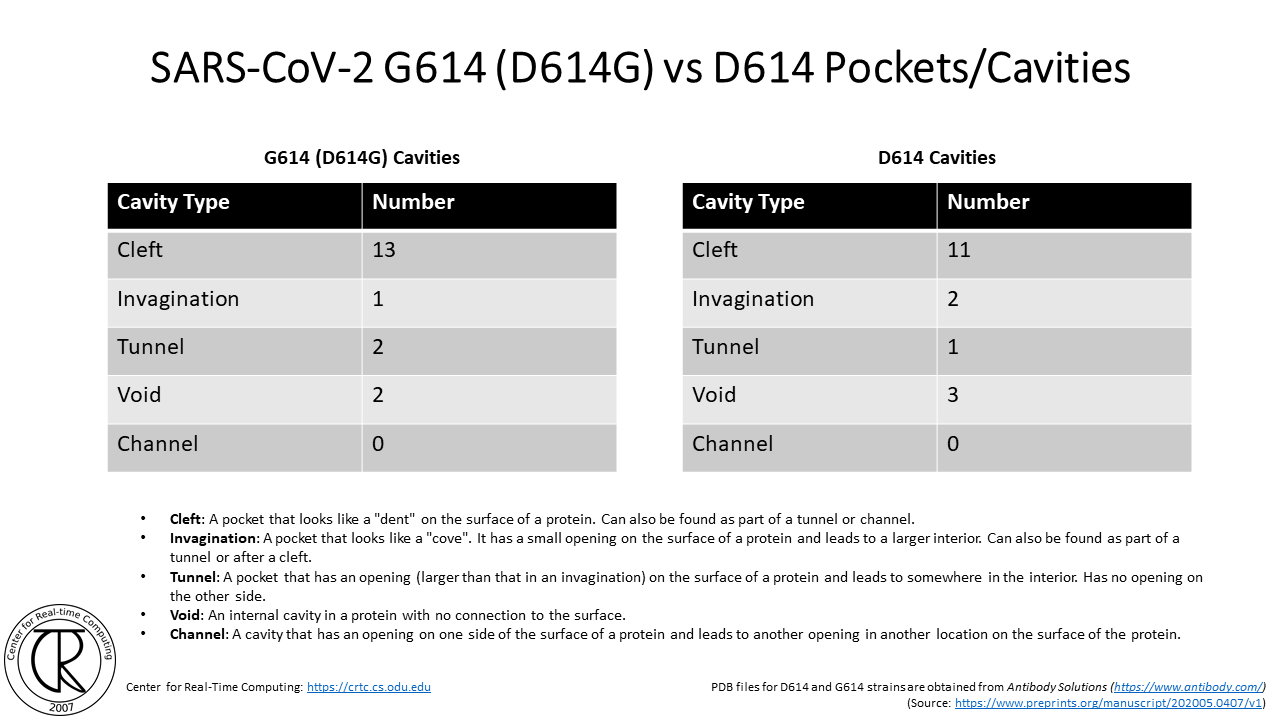
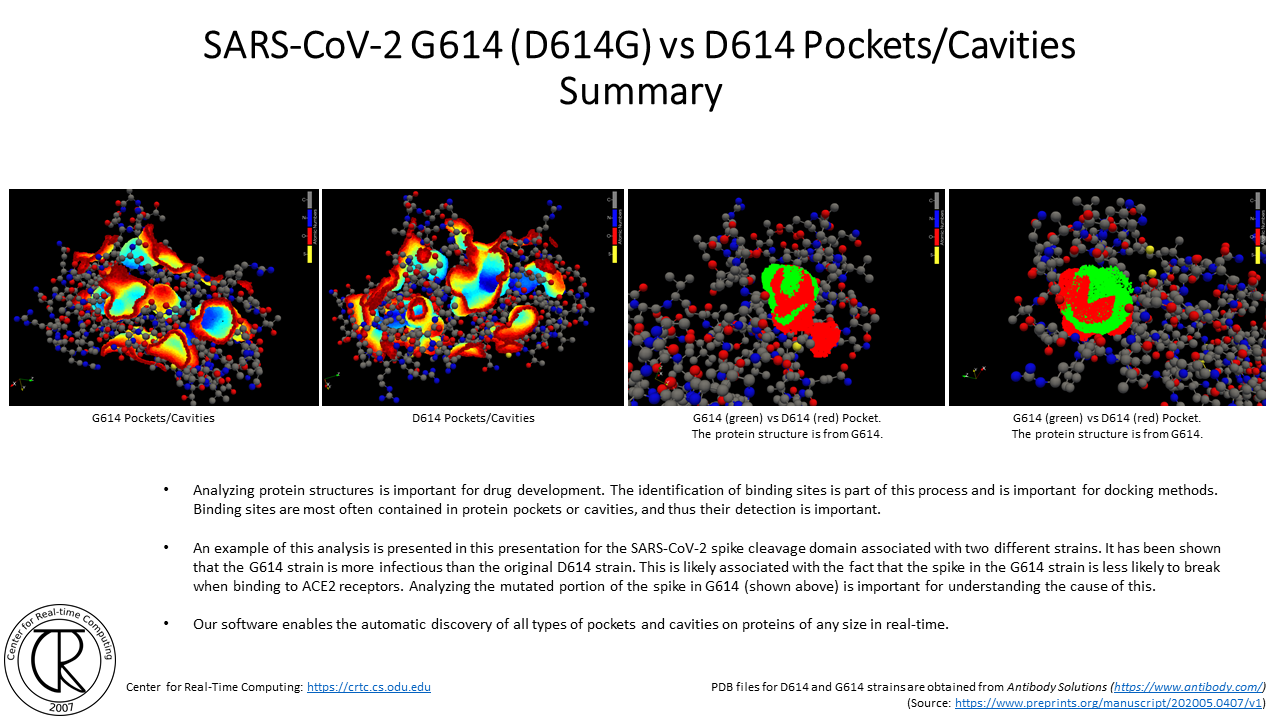
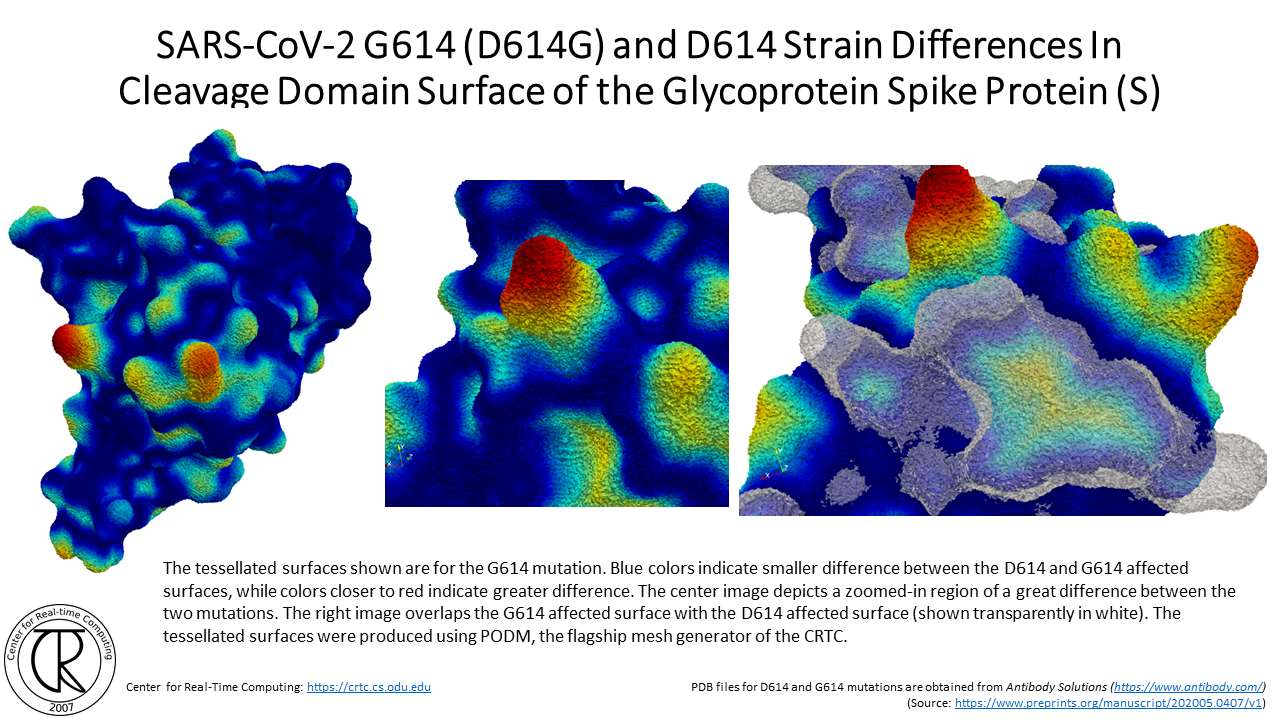
SARS-CoV-2 G614 Spike Cleavage Domain Tunnels (red-blue) Superimposed on D614 Spike Cleavage Domain with D614 Tunnel (purple-orange) using the atomic structure from the original strain
Remark: Notice the difference between the position of new tunnels where the C & O used to be in the original COVID19 strain
To see other videos please click here.
SARS-CoV-2 D614 vs G614 Mutation in Spike Cleavage Domain
To see other slideshows please click here.
Workshops
| DATA SCIENCE ROADMAP TO COMPTON FORM FACTORS OF QUARKS AND GLUONS |
|---|
| Chrisochoides' Real-Time Computing (CRTC) lab in collaboration with Dr. Charles Hyde (ODU), Dr. Simonetta Liuti (UVA), and Dr. David Heddle (CNU) will host a two-day mini-workshop on the roadmap to Compton Form Factors for efficient evaluations of Monte Carlo Simulations. For more information see: DATA_SCIENCE_ROADMAP_TO_COMPTON_FORM_FACTORS_OF_QUARKS_AND_GLUONS |
| CNF HPC Workshop | CNF Imaging Workshop |
|---|---|
| The CRTC lab in collaboration with the Jefferson lab hosted a one-day mini-workshop on Imaging and High Performance Computing (HPC). For more information see: CNF_HPC_Workshop | The CRTC lab in collaboration with the Jefferson lab hosted a two-day mini-workshop on Imaging and Nuclear Femptography. For more information see: CNF_Imaging_Workshop |
Introduction
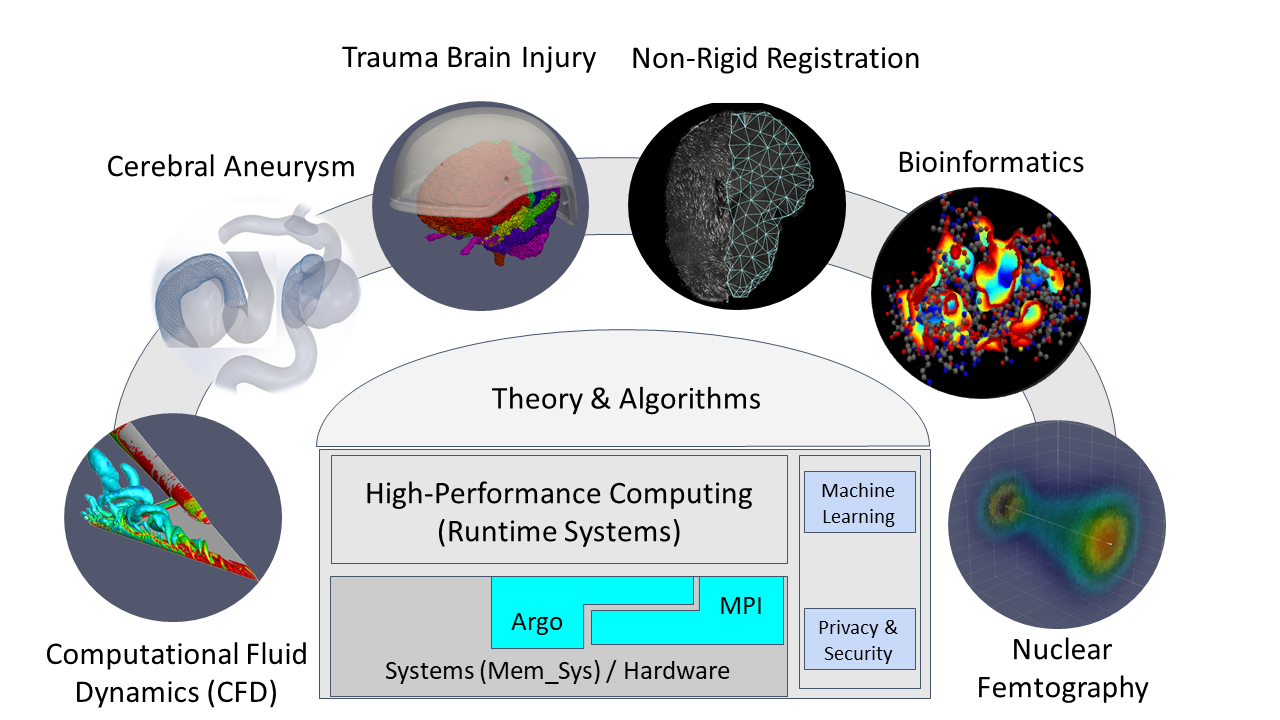
Finite Element (FE) mesh generation is a critical component for many (bio-)engineering and science applications. CRTCLab is developing a novel framework for highly scalable and energy-efficient high-quality mesh generation for the Finite Element Analysis in three and four dimensions. CRTC effort and focus is on research activities that combine domain-and application-specific knowledge with run-time system support to improve energy efficiency and scalability of parallel FE mesh generation codes. Traditionally, parallel FE mesh generation methods and software are developed without considering the architectural features of the supercomputer platforms on which they are eventually used for production. The main reason is the complexity of sequential, and moreover parallel, mesh generation algorithms. As a result, it is too expensive, in terms of labor and time, to customize the performance of parallel mesh generation software for specific supercomputing architectures. CRTC’s basic research is funded from NSF, NASA, NRA, total $2M and target (bio-)engineering applications are: Aerospace Industry Applications and Health Care Industry Applications.
Principles for Building Practical and Scalable Domain-Specific Environment: Adaptive Data-Driven Mesh Generation for Modeling & Simulations
Broader Impact
The proposed activity will have a significant and broader impact on research and education, well beyond the bounds of this specific project and its external collaborators. Other than the direct impact to parallel unstructured mesh generation and runtime systems for supercomputers like Blue Waters, this project will benefit the performance evaluation community. This project will also promote the understanding on CerebroVascular Disease (CVD, or stroke), Image Guided Surgery for Caner, and DBS for Parkisons disease, all of which are leading natural death causes in the US.
Intellectual Merit The Intellectual Merit of this project is summarized in terms of three novel contributions: (1) an understanding of the complex computation, communication, and memory utilization patterns in a telescopic superposition of multiple parallel mesh generation codes, (2) new abstractions to efficiently throttle concurrency and to implement race-to-halt execution models at multiple levels of granularity, while orchestrating intra-and inter-layer memory management, data movement, and load balancing to improve energy-efficiency of memory and interconnect components, and (3) a novel approach to leveraging off-the-shelf and state-of-the-art low core meshing codes for highly parallel simulations.
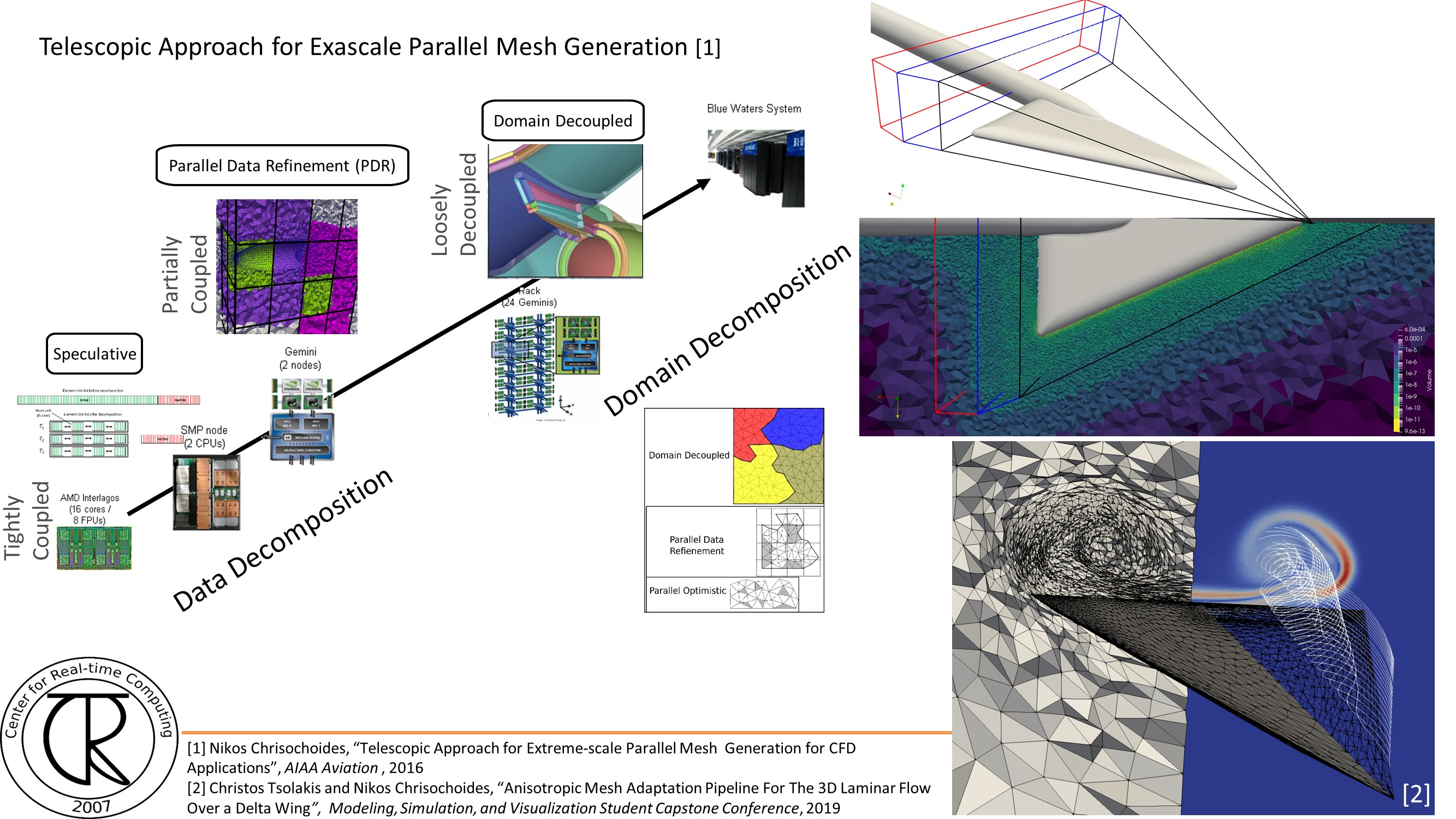
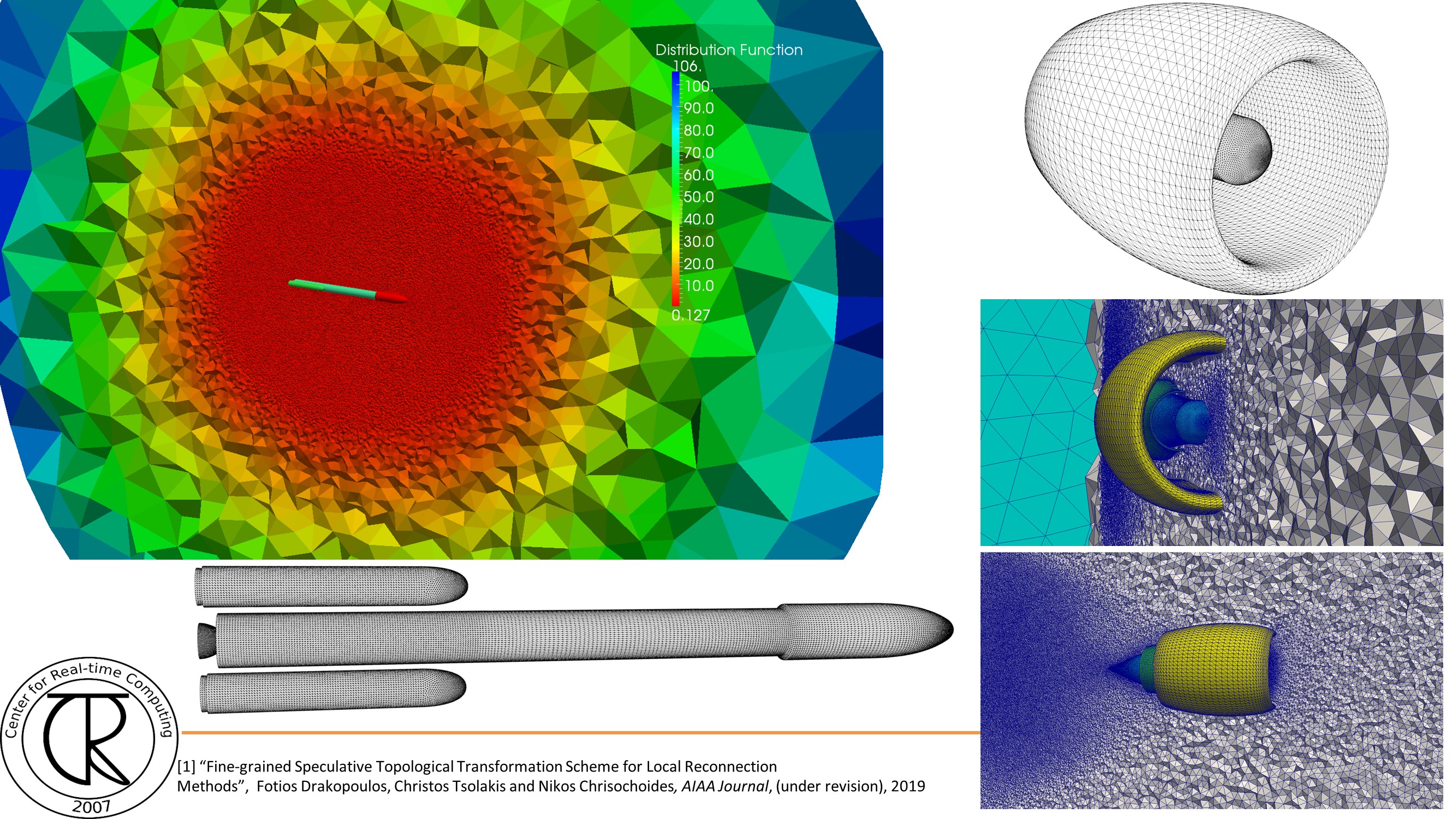
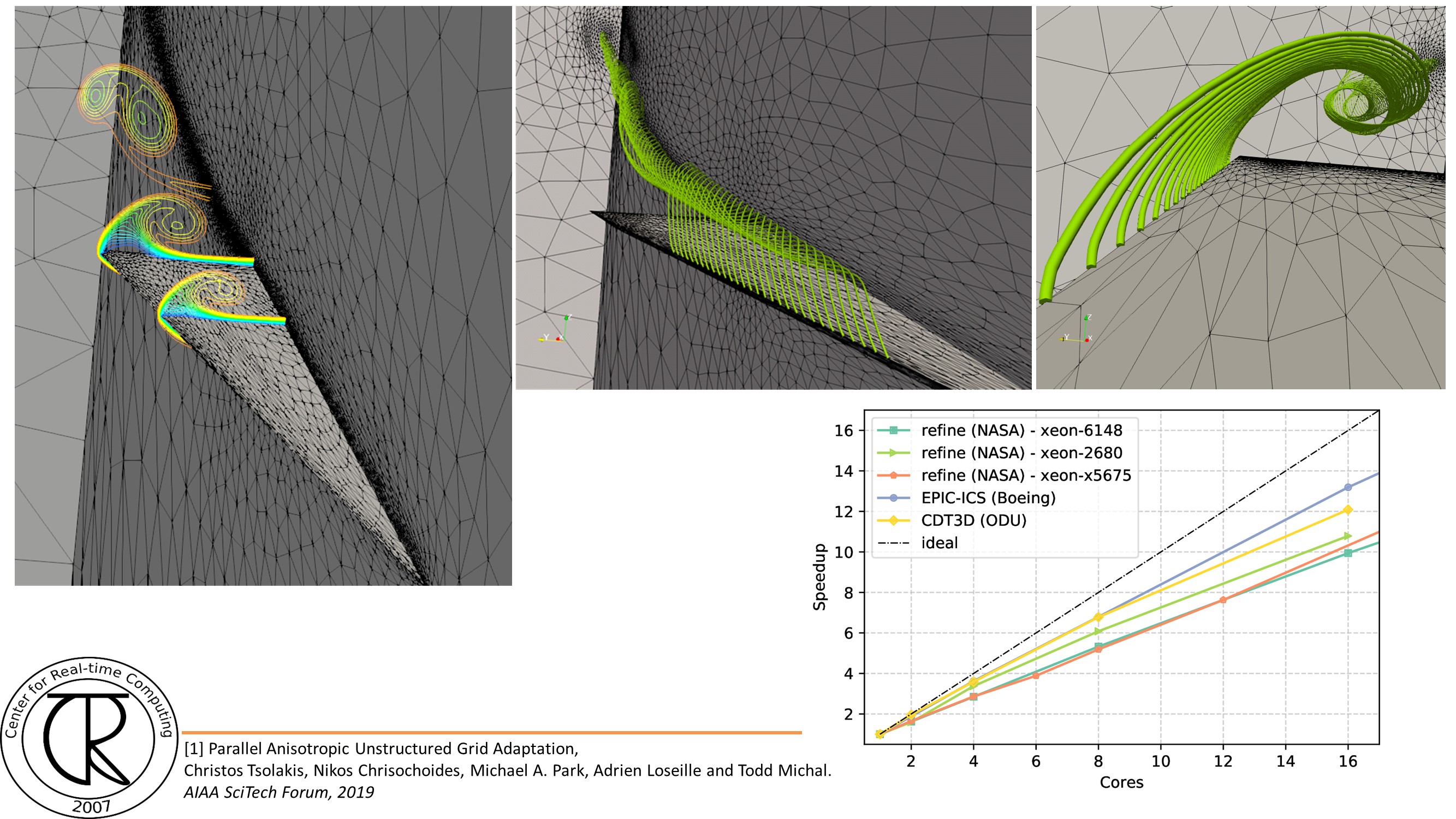
Extreme-Scale Adaptive/Anisotropic Parallel Mesh Generation
- CDT3D Meshes using speculative execution model at the chip level
Overview
Finite Element Mesh Generation is a critical component for many (bio-) engineering and science applications. The goal of this project is to deliver a novel Telescopic framework for highly scalable and energy efficient codes. Domain-and application-specific knowledge and run-time system support are combined to improve accuracy of FE computations.
CRTCLab addresses in depth two pillars: (i) high performance computing software runtime systems and (ii) mesh generation & adaptation based on error-metrics for NASA’s Computational Fluid Dynamics (CFD) Vision 2030. Specifically, CRTC’s focus is on the following three objectives: (1) design a multi-layered algorithmic and software framework for 3D tetrahedral anisotropic parallel mesh generation using state-of-the-art functionality supported by algorithms from AFLR and CRTC’s telescopic approach for parallel mesh generation , (2) develop error-based metrics adaptive anisotropic FE mesh generation methods and (3) design a power-aware parallel runtime software system for extreme-scale adaptive CFD computations including: (i) mesh generation & adaptation, and (ii) consistent error-based metrics for adaptation of any CFD discretizations with localizable error estimates. This is a joined project with NASA/LaRC and MIT.
We have assembled a team of established leaders (see External Collaborators) that are currently developing state-of-the-art work on mesh generation and adaptivity issues relevant to NASA’s CFD 2030 Vision and will broadly impact end-user productivity of users throughout DoD and NASA.
Objectives
- Design a multi-layered algorithmic and software framework for 3D tetrahedral anisotropic parallel mesh generation methods using state-of-the-art functionality supported by methods implemented in AFLR and CRTC’s telescopic approach for parallel mesh generation.
- Development of error-based metrics to drive an anisotropic adaptive process
- Design a power-aware parallel runtime software system for extreme-scale adaptive CFD computations including: (i) mesh generation & adaptation, and (ii) consistent error-based metrics for adaptation of any CFD discretization with localizable error estimates.
Real-time Adaptive Mesh Generation in Medical Image Computing
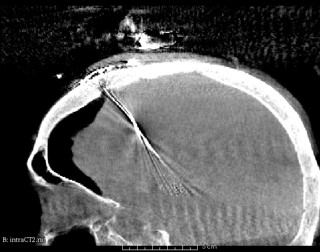
This application is using adaptive (isotropic) mesh refinement to improve (in real-time in the context of image-guided neurosurgery) the accuracy of 3D Physics-Based Deformable Registration of intra-operative MRI with pre-operative MRI.
Overview
- Develop adaptive deformable registration method for Deep Brain Stimulation (DBS). DBS is an effective palliative therapy for patients suffering from Essential Tremor, Parkinson’s disease, and other neurological movement disorders. Modern DBS surgery makes use of stereotactic systems and image guidance to accurately place electrode leads, as well as intra-operative imaging to surveil the location of the leads and guide the surgery. The effectiveness of DBS is directly correlated with the accuracy of DBS electrode lead placement, with more accurate electrode placement leading to better clinical outcomes. This project is a joined with VCU, VA.
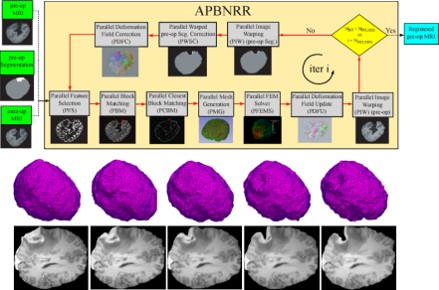
- Develop a new Adaptive Physics-Based Non-Rigid Registration (APBNRR) method developed for image guided brain tumor resection. Existing evaluation indicates that the the registration accuracy and performance of APBNRR are proved sufficient to be of clinical value in the operating room. Malignant gliomas are the most common primary and metastatic brain tumors. Treatment typically includes surgical removal followed by radiotherapy or chemotherapy. Total resection is difficult to achieve during tumor removal because of the infiltrative nature of gliomas and because brain tumors are often embedded in critical functional brain tissue. In image-guided neurosurgery, co-registered pre-operative anatomical, functional, and diffusion tensor imaging can be used to facilitate a maximally safe resection of brain tumors in certain areas of the brain. However, because the brain can deform significantly during surgery, particularly in the presence of tumor resection, non-rigid registration of the preoperative image data to the patient is required. This is a joined project with Harvard Medical School, MA.
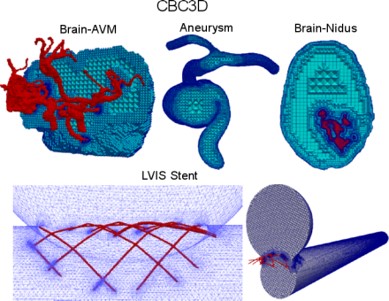
- Test five new available (in the clinic) neurovascular devices that have not been compared before through angiographic washout analysis and accurate micro CT-based tessellation (i.e., mesh or grid reconstruction) and CFD analysis in idealized sidewall aneurysm models. The CFD simulations and angiographic washout analysis will be performed on flow diverter implantations in actual patient geometries. There is a need for robust and easy-to-use Image-to-Mesh (I2M) conversion methods and software that streamline the discretization of such complex vascular geometries with fine structures with the CFD modeling process. In this project we will focus on the development and validation of fully functional I2M conversion algorithms and software (i.e., tools) for complex vascular geometries with fine structures like stents. This is a joined project with Medical School at Stony Brook University, NY.